Therefore, it is unlikely to be that lung-homing defects introduced by terminal differentiation limit the migration of CD4 T cells into granulomas following primary Mtb infection of macaques. Using the intravascular stain approach, we find essentially all Mtb-specific CD4 T cells in granulomas have extravasated across the vascular endothelium into the parenchyma. However, most granuloma CD4 T cells are found within the outer lymphocyte cuff and few localize to the myeloid cell core containing the bacilli. Mtb-specific CD4 T cells in granulomas display a tissue-parenchymal CXCR3 +CX3CR1 −PD-1 hiCTLA-4 + phenotype. Mtb-specific CD4 T cells simultaneously appeared in the airways and blood ∼21–28 days post exposure, indicating that recently primed effectors are quickly recruited into the lungs after entering circulation. Here we examine CD4 T-cell differentiation and lung homing during primary Mtb infection of rhesus macaques. 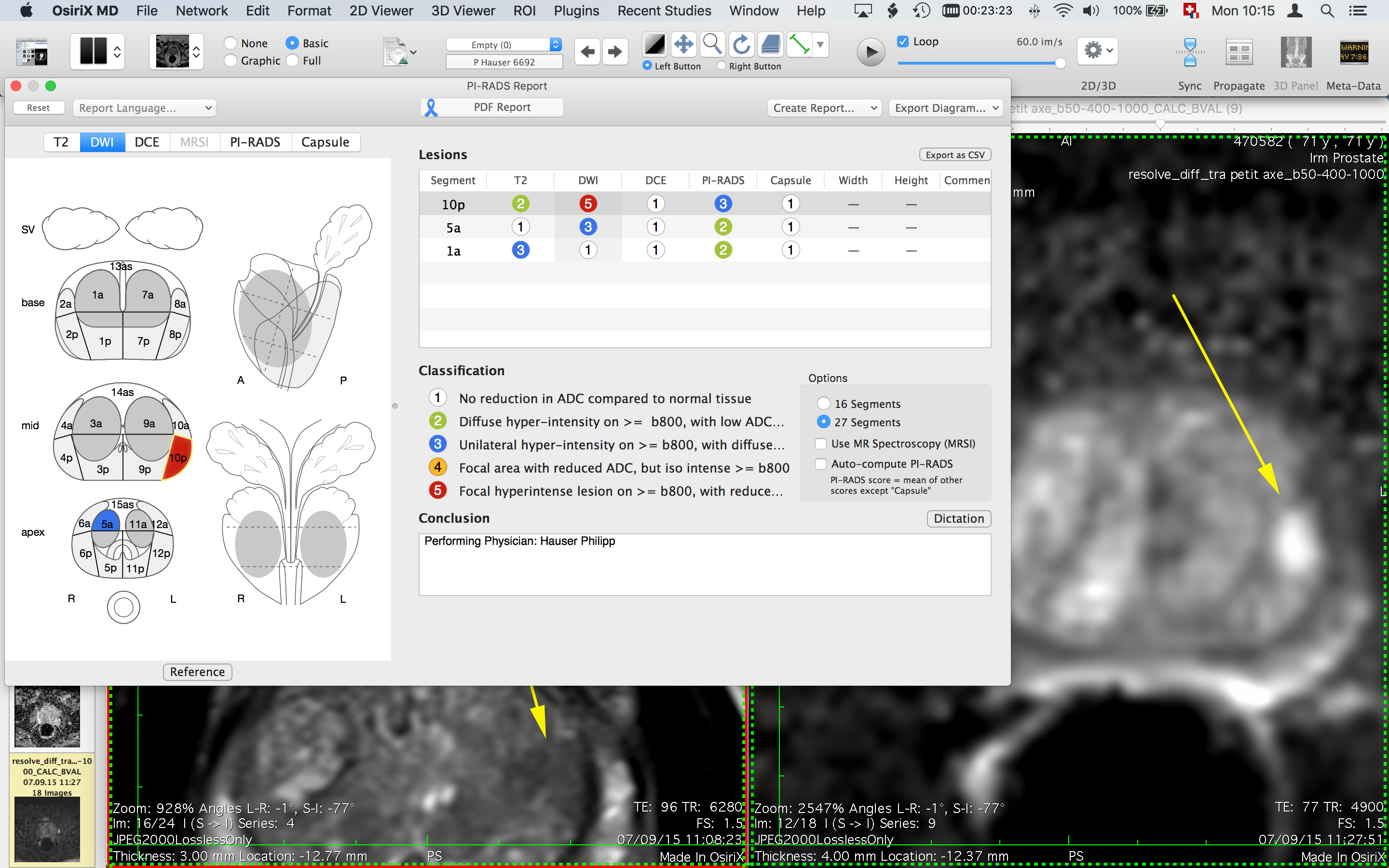
In mice, less-differentiated CXCR3 + CD4 T cells migrate into the lung and suppress growth of Mtb, whereas CX3CR1 + terminally differentiated Th1 cells accumulate in the blood vasculature and do not control pulmonary infection. Protection against Mycobacterium tuberculosis (Mtb) infection requires CD4 T cells to migrate into the lung and interact with infected macrophages. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
